Research
I conduct research in Dr. Tom Huxford's Structural Biochemistry Laboratory at San Diego State University.
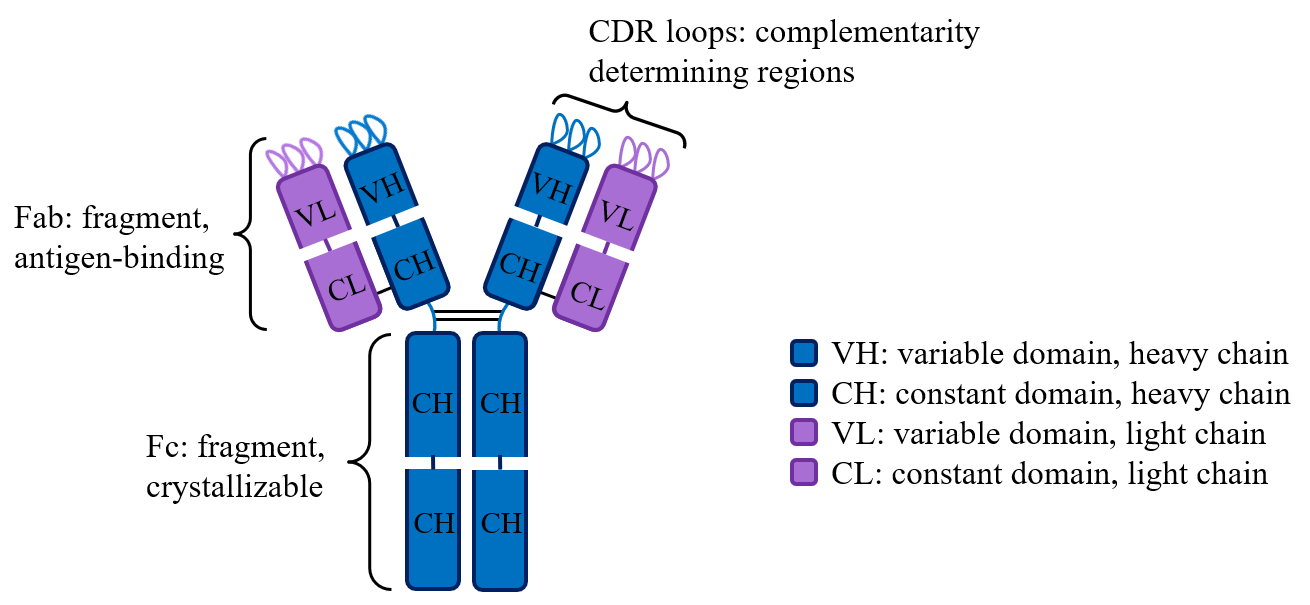
I am currently studying an antibody called Q425. This antibody binds CD4, a transmembrane protein on T cells that human immunodeficiency virus (HIV) binds to before viral infection. I recently used western blots to reveal the recombinant Q425 antibody binding with CD4 in a calcium-dependent manner. Now, I aim to map the epitope on CD4 that Q425 binds to using mass spectrometry.
My previous research project involves the characterization of an anti-dsDNA antibody, 2C10. Due to it’s sequence homology to a previously studied metal binding antibody, we believe calcium increases the binding affinity of 2C10 to its antigen. Determining the binding mode of 2C10 to its antigen may provide valuable insight into the conservation of metal binding antibodies in evolution.
I am currently studying an antibody called Q425. This antibody binds CD4, a transmembrane protein on T cells that human immunodeficiency virus (HIV) binds to before viral infection. I recently used western blots to reveal the recombinant Q425 antibody binding with CD4 in a calcium-dependent manner. Now, I aim to map the epitope on CD4 that Q425 binds to using mass spectrometry.
My previous research project involves the characterization of an anti-dsDNA antibody, 2C10. Due to it’s sequence homology to a previously studied metal binding antibody, we believe calcium increases the binding affinity of 2C10 to its antigen. Determining the binding mode of 2C10 to its antigen may provide valuable insight into the conservation of metal binding antibodies in evolution.